 Michael Collins · Growth-Stage Biotech · Speciality Pharma · Large Cap
Michael Collins · Growth-Stage Biotech · Speciality Pharma · Large Cap
Not there yet.
You will be.
I build the R&D plan and operating model that close that gap — from review to delivery, embedded with your team.
 Michael Collins · Growth-Stage Biotech · Speciality Pharma · Large Cap
Michael Collins · Growth-Stage Biotech · Speciality Pharma · Large Cap
I build the R&D plan and operating model that close that gap — from review to delivery, embedded with your team.
From growth-stage biotech to large cap pharma — I build R&D operating models and plans that earn conviction, then deliver them.

Fortune 500 R&D transformation programmes — proven methodologies applied to every Collins Helvetia engagement.
Your R&D plan will face hard questions from VCs, boards, and partners. I pressure-test every timeline, budget, and risk assumption against industry benchmarks — producing the data-backed narrative that earns investor conviction.
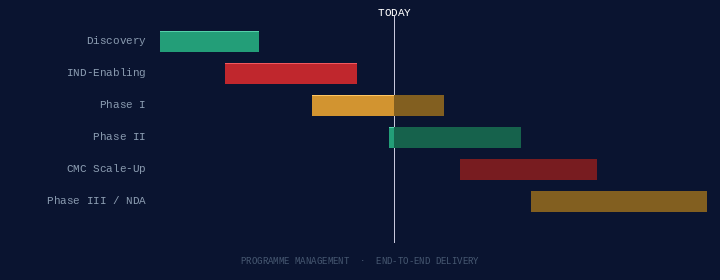
End-to-end programme leadership from early discovery through NDA/MAA — with hands-on CRO and vendor management.

Designing the operating model, decision-making structures, and real-time pipeline visibility that let your organisation move fast and deliver consistently.

Independent assessment of pipeline assets — timelines, regulatory risk, CMC readiness — for buy-side decisions.
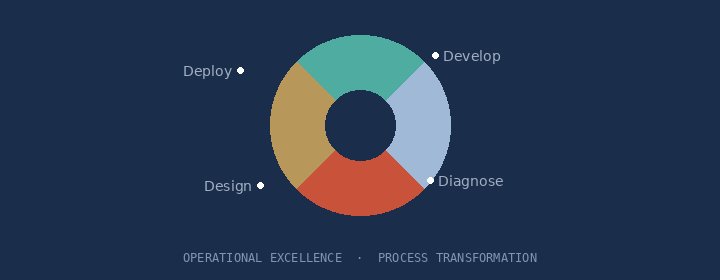
Process design and operating model transformation that embeds efficiency and accountability across R&D.
Practical AI adoption in R&D workflows — automated risk prediction, intelligent portfolio analytics, digital PM tools.
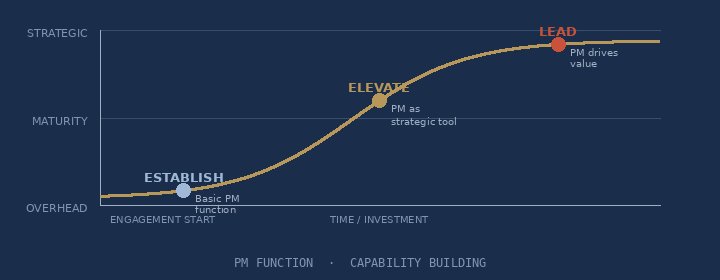
Designing world-class PM functions from the ground up — PM Academies, maturity benchmarking, structured capability pathways.

Structured discovery — understand your pipeline, culture, and the core challenge before recommending anything.
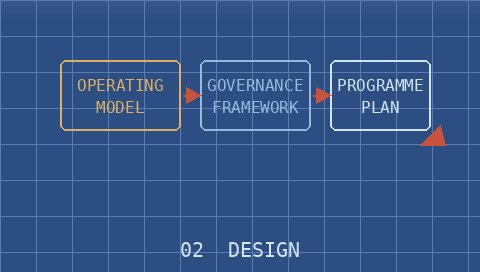
Co-develop a pragmatic, fit-for-purpose solution grounded in your context, not off-the-shelf templates.
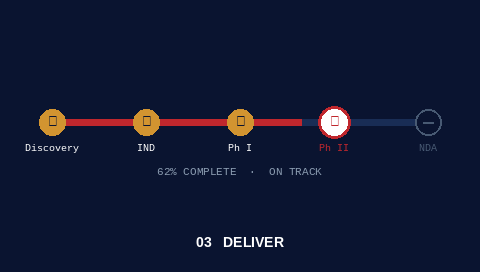
Hands-on execution alongside your team. I hold accountability for outcomes, not just recommendations.
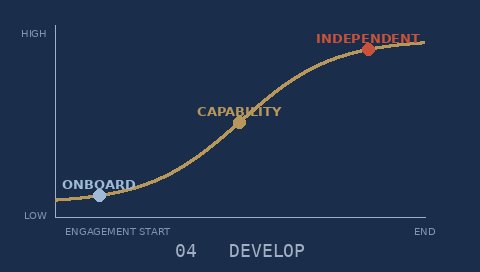
Knowledge transfer so your organisation owns the solution — no ongoing dependency on external support.


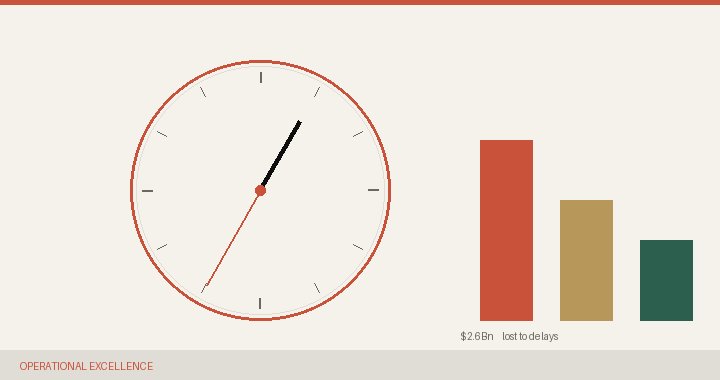
12–17 months of development time is spent waiting for governance meetings — not experiments, not trials.
Read on LinkedIn →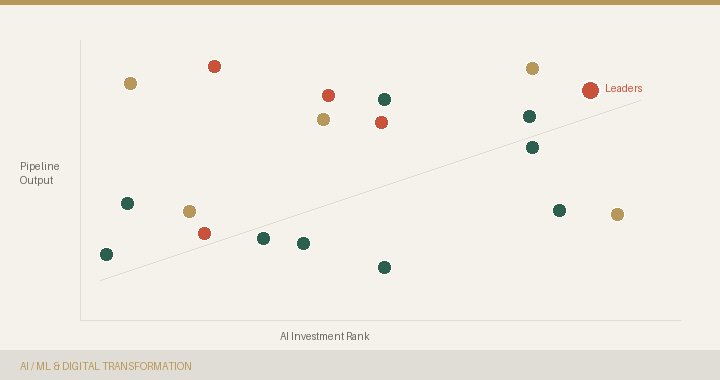
The biggest AI investors in pharma are struggling to translate technology into molecules. An analysis of what separates the leaders.
Read on LinkedIn →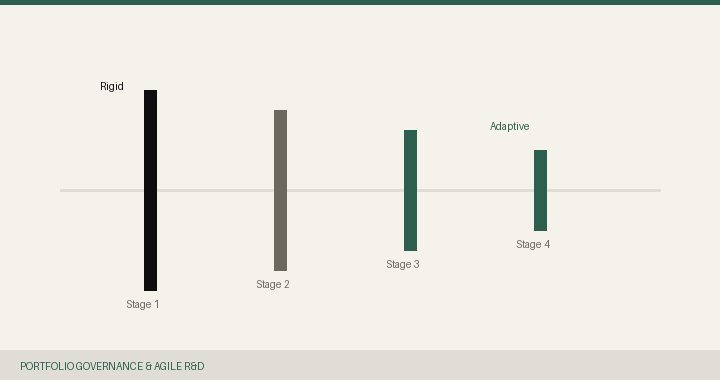
Stage-gate processes were designed for predictability. Drug development is not predictable.
Read on LinkedIn →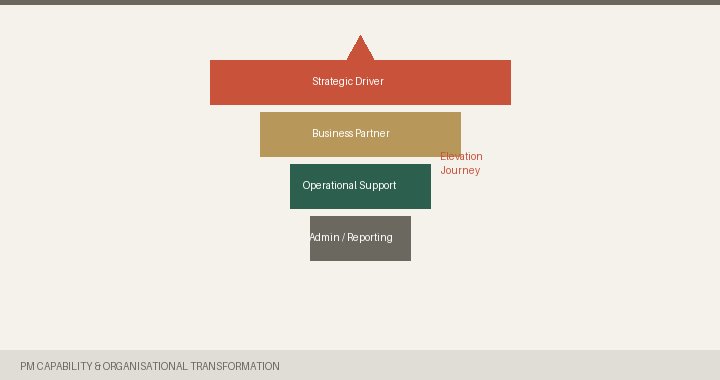
Most R&D PM functions are built to report, not to lead. Here is how to change that.
Read on LinkedIn →"Every engagement begins with a no-obligation 30-minute call to establish fit and scope."
Book a Discovery Call →